PFAS Treatment
Solutions
PFAS in Drinking Water: Protecting Your Community Starts Here
Per- and Polyfluoroalkyl Substances (PFAS), also called “forever chemicals,” are now among the most tightly regulated contaminants in water. Carver Water partners with municipalities, utilities, and businesses to meet new EPA standards and safeguard public health with proven PFAS removal solutions.
PFAS Treatment
Solutions
PFAS in Drinking Water: Protecting Your Community Starts Here
Per- and Polyfluoroalkyl Substances (PFAS), also called “forever chemicals,” are now among the most tightly regulated contaminants in water. Carver Water partners with municipalities, utilities, and businesses to meet new EPA standards and safeguard public health with proven PFAS removal solutions.
What Are PFAS and Why Do They Matter?
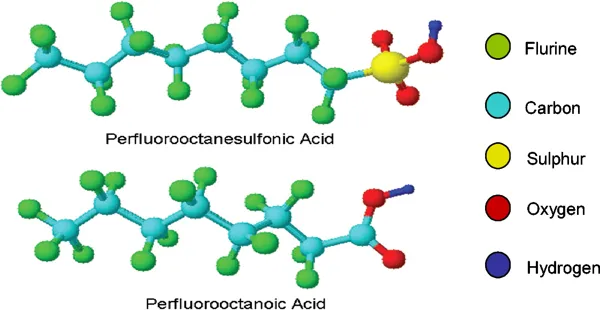
PFAS are a class of over 4,700 synthetic chemicals found in firefighting foams, non-stick cookware, food packaging, textiles, and industrial waste. Nicknamed “forever chemicals” because of their resistance to natural breakdown, PFAS accumulate in water, soil, and even human bloodstreams.
What Are PFAS?
PFAS (Per- and Polyfluoroalkyl Substances) are a large class of over 4,700 synthetic chemicals designed to resist heat, oil, stains, grease, and water.
They are commonly found in:
Firefighting foams
Non-stick cookware
Food packaging and takeout containers
Stain-resistant carpets and textiles
Industrial coatings, paints, and waxes
Why they matter: PFAS are called “forever chemicals” because the fluorine-carbon bond is one of the strongest in chemistry, making them extremely resistant to natural breakdown. Once released, they remain in the environment indefinitely.

How PFAS Enters the Body
PFAS exposure happens through eating, drinking, and even breathing:
By Eating: Food packaging, cookware, contaminated produce, and fish from polluted waters.
By Drinking: The primary concern—PFAS enters groundwater and surface water from manufacturing waste, wastewater plants, or firefighting foam.
By Breathing: Household items like treated carpets and clothing can release PFAS into indoor air.

Health Risks Linked to PFAS Include:
Cancer: Kidney and testicular cancers
Thyroid disorders and hormone disruption
Pregnancy complications including hypertension
High cholesterol and cardiovascular risks
Immune suppression leading to weaker defenses against illness
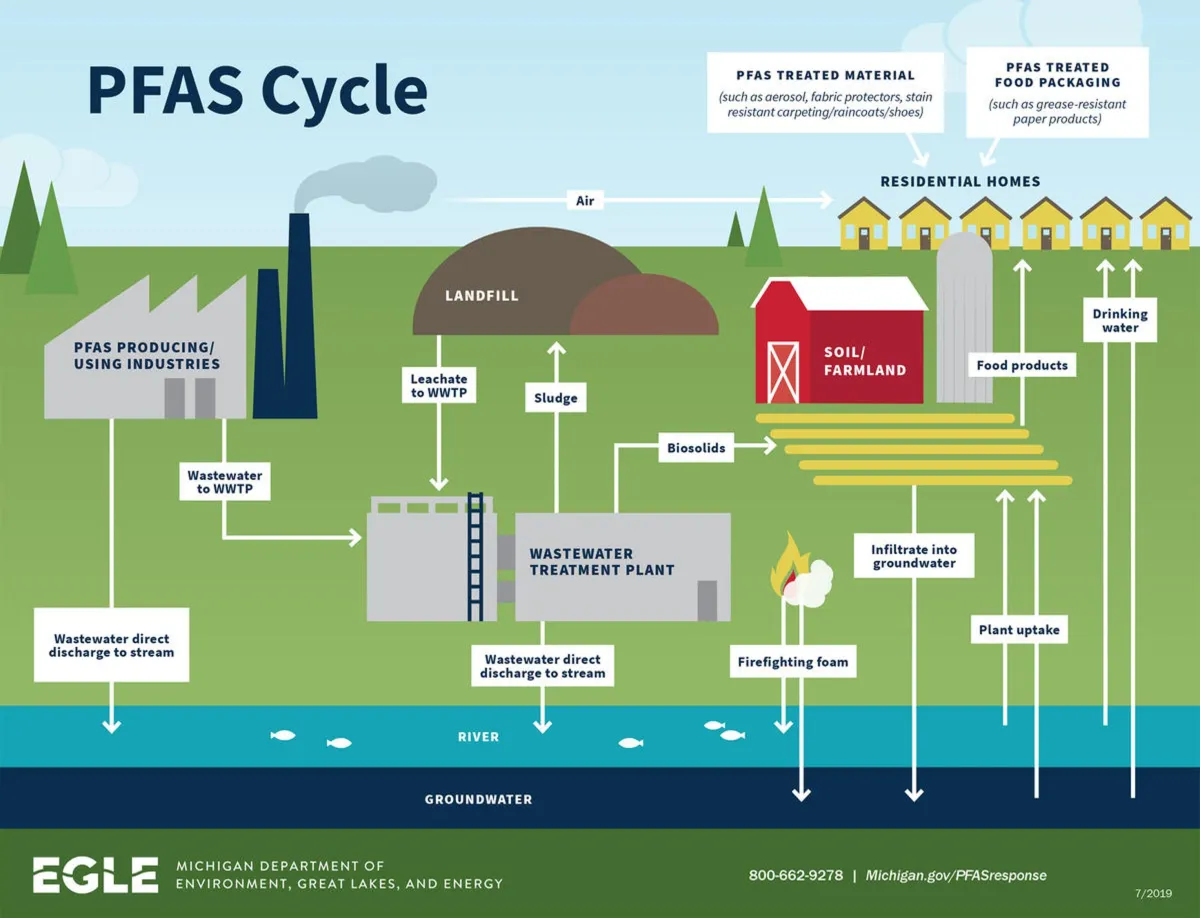
PFAS In The Environment
Once released, PFAS contaminate groundwater and rivers, threatening drinking water supplies and aquatic life for decades.
PFAS contamination isn’t limited to one community—it’s widespread:
More than 1,400 industrial and commercial sites in the U.S. are contaminated
Over 140 Department of Defense sites are affected
A 2020 report projects $3 billion annually will be spent by 2030 on PFAS remediation technologies
Public awareness is also growing, fueled by media coverage, social pressure, and tightening regulations. For utilities and industries, this means urgent action is required.

Interested In Sampling Your Well or Tap water?
If you are interested in sampling your private residential drinking water well or tap water for PFAS
You may also be interested in the following resources:

PFAS and EPA’s New Drinking Water Regulations
In 2024, the U.S. Environmental Protection Agency set strict Maximum Contaminant Levels (MCLs) for for six key PFAS chemicals—including PFOA, PFOS, PFHxS, PFNA, and GenX (HFPO‑DA)—as well as a Hazard Index for certain mixtures Source.
For municipalities and water plants, compliance isn’t optional—it’s law. Non-compliance can result in regulatory penalties and, more importantly, risks to public health.
Key Details:
Stakeholders
Requirements & Implications
Municipalities / Water Systems
Must begin testing by 2027, then treat and comply by 2029. Public disclosure and notification are required if levels exceed MCLs
Industrial Facilities
Must monitor discharges and may need to upgrade treatment systems to avoid pollution liability under the Safe Drinking Water Act and related regulations
Funding & Support
The EPA is offering grants and financial assistance, including up to $1 billion to support testing and treatment infrastructure
Let's tackle this regulatory issue together.
PFAS Removal Technologies
Granular Activated Carbon (GAC)
Strengths: Effective for long-chain PFAS, relatively low cost, widely used in municipal plants.
Limitations: Less effective for short-chain PFAS (e.g., GenX), frequent media replacement needed, hazardous waste disposal challenges.

Ion Exchange (IX)
Strengths: Removes both long- and short-chain PFAS efficiently; effective for polishing treated water.
Limitations: Higher cost, resin fouling, and disposal of PFAS-laden brine.

Nanofiltration (NF) & Reverse Osmosis (RO)
Strengths: Highly effective for comprehensive PFAS removal, scalable for municipal and industrial use, also removes salts and other contaminants.
Limitations: High energy use, membrane fouling, concentrated PFAS brine disposal.

Advanced Oxidation (AOP) & Electrochemical Oxidation
Strengths: Potential to destroy PFAS molecules instead of just separating them, eliminating brine waste.
Limitations: High energy costs, incomplete breakdown risks, and formation of toxic byproducts.
DRACO™ (Wastewater Sludge Densification)
Purpose: Treat municipal or industrial wastewater sludge high in organics to create a denser sludge that concentrates PFAS mass, improving overall incineration/destruction efficiency and reducing logistics.
Use cases: WWTP digesters/DAF/clarifier solids, industrial sludges.
Not for: Drinking water or dissolved-phase PFAS removal.
Learn more → /technologies/draco

We Bring Municipal and Industrial
PFAS Removal Solutions
Solution 1 —
Dissolved PFAS in
Drinking Water

Choose one of: GAC, IX, or RO based on targets, water chemistry, and lifecycle cost.
Solution 2 —
PFAS in
Wastewater Discharge

Choose one of: GAC, IX, or RO based on targets, water chemistry, and lifecycle cost.
Solution 3 —
PFAS in
Wastewater Sludge

Use DRACO™ to thicken/condense sludge into a denser residual so offsite thermal destruction is more efficient.
Looking for an in-depth technology recommendation? →
Industries we serve
Drinking Water Utilities
Treatment Technology: GAC or IX; RO for ultra-low targets or reuse.
Support: Pilots, rate case support, public communications kits.
Industrial & Municipal Wastewater
Treatment Technology: IX or RO to prevent PFAS discharge.
Solids Handling: DRACO™ to densify sludge for efficient, compliant destruction. Learn More About DRACO → /wastewater/DRACO
Ask for a tailored solution→
Frequently Asked Questions
Q1: What exactly are PFAS and why are they called “forever chemicals”?
PFAS (Per- and Polyfluoroalkyl Substances) are a class of over 4,700 synthetic fluorinated compounds. Their key feature is a fluorine-carbon (C–F) bond, one of the strongest in chemistry, which makes PFAS highly resistant to heat, chemical breakdown, and environmental degradation. This durability means PFAS persist indefinitely in water, soil, and living organisms—hence the name “forever chemicals.”
Q2: Which PFAS compounds are regulated under the new EPA rules?
In 2024, the EPA finalized Maximum Contaminant Levels (MCLs) for several PFAS:
PFOA and PFOS: Set at 4 parts per trillion (ppt) each.
GenX (HFPO-DA) and PFBS: Regulated individually at higher ppt thresholds.
PFNA and PFHxS: Addressed in a combined hazard index approach.
These levels are extremely low, reflecting the toxicity of PFAS even in trace concentrations.
Utilities must monitor and treat water to meet these limits.
Q3: How do PFAS typically enter drinking water systems?
PFAS contamination occurs through several pathways:
Industrial discharges from manufacturing plants.
Wastewater treatment plant effluent, which returns PFAS-laden water to rivers.
Firefighting foam (AFFF) runoff from military bases and airports.
Landfill leachate and biosolids applied to agricultural fields.
Atmospheric deposition from PFAS released through factory emissions.
Once PFAS reach surface water or groundwater, they spread widely and infiltrate municipal supplies.
Q4: Which PFAS treatment technologies are most effective?
There is no single “perfect” technology, but leading options include:
Granular Activated Carbon (GAC): Effective for long-chain PFAS like PFOA and PFOS, but less efficient for short-chain compounds such as GenX.
Ion Exchange (IX): Strong-base anion resins can remove both long- and short-chain PFAS, but resin fouling and PFAS-concentrated brine disposal remain challenges.
Reverse Osmosis (RO) and Nanofiltration (NF): RO provides comprehensive removal of nearly all PFAS, while NF is more energy-efficient but less effective on short-chain PFAS. Both generate brine waste streams requiring careful disposal.
Advanced Oxidation Processes (AOP) and Electrochemical Oxidation: These methods can potentially degrade PFAS at the molecular level, but they are energy-intensive and still emerging technologies.
Carver Water often engineers integrated systems (e.g., GAC + RO + IX polishing) to balance cost, efficiency, and waste management.
Q5: What happens to PFAS after treatment—are they destroyed?
This depends on the method:
Adsorptive methods (GAC, IX): PFAS are captured in the media, which then becomes hazardous waste requiring regeneration or high-temperature incineration.
Membrane methods (RO, NF): PFAS are concentrated into a brine waste stream, typically 15–25% of the treated water volume. Brine disposal (deep well injection, evaporation ponds) is costly and tightly regulated.
Destructive methods (AOP, Electrochemical): Aim to break down PFAS into less harmful byproducts. However, incomplete degradation can lead to toxic intermediates, and these technologies are still under optimization.
Q6: How big is the PFAS treatment market?
According to Bluefield Research, U.S. utilities will spend over $3 billion annually by 2030 on PFAS remediation technologies. Drivers include:
-Growing public health awareness.
-New state and federal mandates under the Safe Drinking Water Act.
-Federal grants funding PFAS technology pilots.
-Legal liabilities faced by manufacturers and utilities.
This makes PFAS the next major challenge for U.S. water utilities.
Q7: What are the biggest challenges with PFAS compliance?
Ultra-low regulatory limits (parts per trillion) require highly sensitive monitoring and treatment.
Short-chain PFAS removal remains technically difficult.
Disposal of PFAS waste (spent media, brine) is costly and environmentally sensitive.
High operational costs for advanced technologies like RO and AOP.
Uncertainty around future regulation—EPA is expected to expand its list of regulated PFAS compounds.
Carver Water’s role is to help utilities choose the right mix of technologies, secure funding, and manage ongoing compliance.
👉 Still have questions?
We're Here To Help!
PFAS isn’t going away—but your community doesn’t have to face it alone.
Let's remove PFAS together!

Innovation
Fresh, creative solutions.

Integrity
Honesty and transparency.

Excellence
Top-notch services.

FOLLOW US
TECHNOLOGY & SERVICES
ABOUT US
CUSTOMER CARE
LEGAL
Copyright 2026. Carver Water . All Rights Reserved.
